 A research paper co-authored by Professor Niamh Nowlan, UCD School of Mechanical and Materials Engineering, has been featured in the journal Science Advances.
A research paper co-authored by Professor Niamh Nowlan, UCD School of Mechanical and Materials Engineering, has been featured in the journal Science Advances.
The study revealed how mechanical forces caused by foetal movements drive skeletal development in the embryo. Its findings indicate that an ion channel called TRPV4 may be a valuable target in future treatments for abnormalities of paediatric skeletal development.
Led by first author Dr Nidal Khatib, Postdoctoral Researcher at Imperial College London, and Professor Niamh Nowlan, Full Professor of Biomedical Engineering at UCD, the study was funded by a European Research Council (ERC) Starting Grant.
Professor Nowlan said: "This research identifies the TRPV4 channel as playing a key role in translating mechanical signals from a baby's movement in the womb to healthy development of the skeleton. The channel could in the future form part of a therapy that would reduce the effects of reduced or restricted foetal movements on skeletal development in babies."
Foetal movements are a critical sign of a baby’s health and development. Such movements are also important for development of the baby’s bones and joints. When a baby doesn’t move enough, or their movements are restricted in some way, the shapes of their joints don’t form correctly, leading to conditions such as developmental dysplasia of the hip (where the hip joint is unstable or dislocated) or arthrogryposis, where multiple joints are angled abnormally.
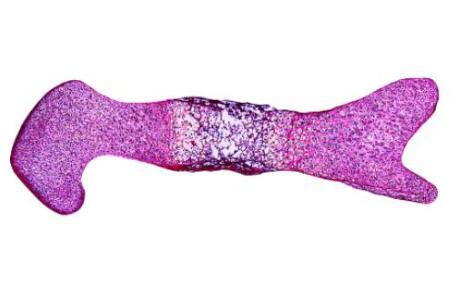
There is a link between the mechanical forces caused by foetal movements and the processes by which the skeleton takes its shape, but the mechanisms underlying this relationship are unknown. The study found that a particular ion channel called TRPV4 (transient receptor potential cation channel subfamily V member 4) is involved in the response of the growing skeleton to the mechanical forces caused by foetal movements. The authors discovered this link by blocking activity of TRPV4 in embryonic mouse limbs, and showing that the normal response of the tissues to mechanical loading was eliminated. The authors also showed that activation of TRPV4 by mechanical loading affects proliferation of cells and the production of matrix in the cartilage, both of which affect growth of the joint.
When the gene which codes for the TRPV4 protein is mutated, a range of diffe rent severe skeletal conditions can occur including lethal metatropic dysplasia, spondylometaphyseal dysplasia (dwarfism), and autosomal dominant brachyolmia.
rent severe skeletal conditions can occur including lethal metatropic dysplasia, spondylometaphyseal dysplasia (dwarfism), and autosomal dominant brachyolmia.
This study is the first to demonstrate that TRPV4 activity in the developing skeleton is closely linked to the mechanical loading from foetal movements. The findings of this research indicate that TRPV4 may be a valuable target for future therapeutic disease modifying drugs for abnormalities of paediatric skeletal development, particularly when foetal movements are reduced or restricted.
Read the paper here: www.science.org/doi/10.1126/sciadv.ade2155
Image: "Revealing how mechanical forces drive skeletal development" as featured in Science Advances.