Haematopoetic Stem Cell Transplantation (HSCT): What We Can Do to Fight Complications
Since 1968, the year in which the first non-twin (allogeneic) bone marrow transplant was performed successfully1, HSCT has been used as the only curative option for certain leukaemias and other disorders such as primary immunodeficiency. Recently, the increasing use of peripheral blood stem cells for autologous transplant has been reported. Compared to conventional bone marrow transplantation, peripheral blood stem cell transplants have a lower regimen-related and transplant-related mortality2. The procedure for both bone marrow and blood sourced HSCT is illustrated in Figure 1.
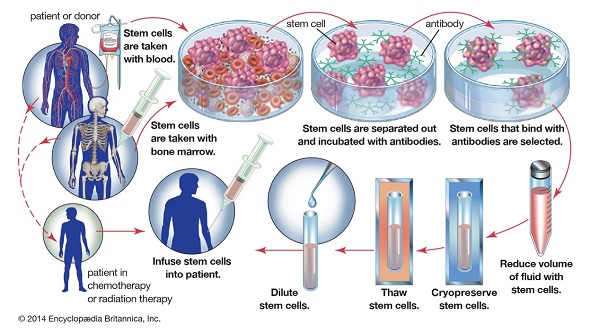
Figure 1. HSCT procedure (https://www.britannica.com/media/full/topic/240689/123191)
Despite the great success of allogeneic HSCT, severe, sometimes life-threatening inflammatory complications can occur due to the differences in major and minor transplantation antigens between donor and patient. Endothelial damage (damage to the lining cells of blood vessels) accounts for most of these complications, such as graft versus host disease (GVHD) and thrombotic angiopathies (TMA). The endothelial injury pathway that explains how HSCT can lead to TMA is illustrated in Figure 2. Ways to prevent transplant-related complications, such as TMA, need to be elucidated and will require personalized precision medicine because the clinical course of these complications is very unique to the patient.
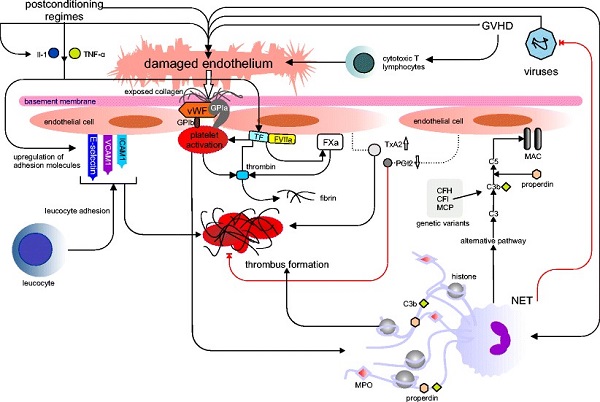
Figure 2. Endothelial injury pathways in TMA. Extrinsic factors from haematopoietic stem cell transplantation (HSCT). 2
White cells from the donor specifically attack the recipients’ endothelial cells, as our group has shown previously 3. By culturing these donor white cells with patient-specific endothelial cell lines generated in the lab, we aim to quantify the risk of endothelial damage for individual donor-recipient pairs in a paediatric cohort of patients. The procedure by which we want to generate specific cell lines differentiates so-called mesenchymal stem cells (MSCs) from bone marrow, into endothelial cells, and transforms these into robustly growing tissue culture cells. These culture experiments can then be used to identify which patient-donor pairs are most likely to suffer severe endothelial damage. Based on this information, high risk patient donor pairs can be prescribed a prophylactic drug regimen to protect the endothelium during the transplant period.
We are confident that this personalised medicine approach will allow a higher positive outcome rate for paediatric and adult patients alike. This work is funded by a grant from the National Children’s Research Center. This grant allows us to closely collaborate with clinicians from Children’s Health Ireland at Crumlin, Dublin.
Figure 3. A 4-year-old boy suffering from primary immunodeficiency in a special hospital bed waiting for HSCT opportunity . (Photo generated from https://kknews.cc/zh-sg/health/kab885q.html)
When I was still an intern in Xiangya Hospital, Changsha, China, I saw many children suffering from immunodeficiency caused by leukaemia. They had to stay in special hospital beds (figure 3), as even small infections could cause severe damage to their bodies. HSCT is a real hope for them. At that time, I couldn’t imagine I could complete my PhD in the field of stem cell therapy and immune reactions against endothelial cells. Now with this opportunity at UCD, I can contribute to this field and be a proud stem cell scientist.
Last but not the least, keep calm and Stemmy will save the world!

References:
- https://www.fredhutch.org/en/treatment/long-term-follow-up/FAQs/transplantation.html
- Hosing, Chitra. "Hematopoietic stem cell mobilization with G-CSF." In Stem Cell Mobilization, pp. 37-47. Humana Press, Totowa, NJ, 2012.
- Seaby, Eleanor G., and Rodney D. Gilbert. "Thrombotic microangiopathy following haematopoietic stem cell transplant." Pediatric Nephrology33, no. 9 (2018): 1489-1500.
- Eissner, Günther, Isabel Hartmann, Altug Kesikli, Ernst Holler, Silvia Haffner, Tanja Sax, Carolin Schray, Bruno Meiser, and Bruno Reichart. "CD4+ CD25+ FoxP3+ regulatory T cells enhance the allogeneic activity of endothelial-specific CD8+/CD28− CTL." International immunology23, no. 8 (2011): 485-492.
Abbreviations:
CFH: complement factor H
CFI: complement factor I
GVHD: graft versus host disease
HSCT: haematopoetic stem cell transplantation
IL-1: interleukin 1
ICAM-1: intercellular adhesion molecule 1
MSCs: mesenchymal stem cells
MAC: membrane attack complex
MPO: Myeloperoxidase
MCP: Membrane Cofactor Protein, a.k.a CD46
NETs: neutrophil extracellular traps
PGI2: prostaglandin I2
TMA: thrombotic angiopathies
TNF-α: tumour necrosis factor alpha
TF: tissue factor
TxA2: thromboxane A2
vWF: von Willebrand factor
VCAM-1: vascular cellular adhesion molecule 1

