Insights to molecular regulation of cilium structure and composition
- Cilia are crucial to sensation, cell-cell communication and movement
- Defects in cilia cause a wide range of developmental diseases
The surfaces of most cell types are covered by hair-like cilia that play critical roles in cell and fluid motility, environment sensing, and cell-cell communication (signalling). New research from University College Dublin shows that cells use a common set of proteins in different ways to build cilia with distinct structures and functions.
This shared set of proteins is also shown to have distinct roles in restricting particular ‘gating’ components to the base of cilia that control the molecular composition of the structure. Defects in cilia cause a wide range of developmental diseases (ciliopathies) that affect nearly every organ system in the human body. Examples include polycystic kidney disease, nephronophthisis, retinitis pigmentosa, Bardet–Biedl syndrome, Joubert syndrome, and Meckel-Gruber syndrome.
Associate Professor Oliver Blacque and Dr Noemie Scheidel from UCD Conway Institute and UCD School of Biomolecular & Biomedical Science are investigating the molecular pathways underpinning cilium biology and disease.
“In this study, we wanted to know how cells use their common intraflagellar transport (IFT) machinery to build cilia with different shapes and functions. We also wanted to determine if IFT and the ‘gating’ machinery at the ciliary base interact in some way, given that both regulate cilium formation, composition and signalling output”, said Associate Professor Blacque.
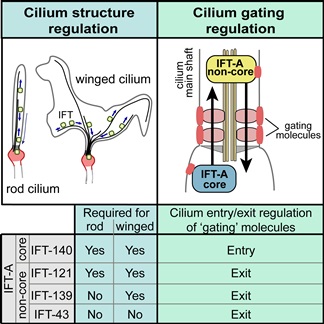
Image caption: IFT-A core and non-core proteins are differentially required for building cilia with different shapes and for transporting gating molecules into or out of cilia.
The intraflagellar transport system occurs along ciliary microtubules (train tracks), employing molecular motors (engines) and at least two types of cargo adaptor complexes (wagons), known as the IFT-A and IFT-B protein complexes.
Dr Noemie Scheidel carried out the experiments in this study using the nematode (roundworm) model organism, C. elegans. Using genetic and fluorescent imaging techniques, she was able to establish the requirements of different IFT-A genes in building cilia of distinct structure and function.
Dr Scheidel also used the same gene ‘knockouts’ and fluorescent versions of ‘gating’ components to investigate if IFT-A genes control how these molecules are summoned to the cilium base.
“Our new work has revealed that IFT-A complexes are deployed in distinct ways in different cell types and that IFT complexes are involved in ensuring that ‘gating’ molecules are correctly restricted to their zone of function at the cilia base.
The next step is to bring this work into human cells to see if the findings are like what we found in nematodes”, said Dr Blacque.
He adds, “Ultimately, we want to understand how human cells regulate cilium formation and composition, and provide new research avenues towards ciliary disease understanding and possible therapy”.
The research is published in the current issue of the journal, Current Biology and is available online - https://doi.org/10.1016/j.cub.2018.08.017
Social Media Links